There are six noble gases and they make up the last column or 18th group of the periodic table. They are called noble because they do not easily combine with other elements to make chemical compounds. This makes them useful in situations where you do not want chemical reactions to occur. For example argon is put into light bulbs to prevent oxidation of the filament. Argon is also used in welding to prevent oxides from forming and contaminating the weld.
SAM postulates that as a nuclet grows it reaches the carbon state where it has a valence of +4/-4 and is in a perfect icosahedron. In order to grow the icosahedron must be distorted slightly. Due to limitations imposed by dense packing the nuclet can grow in only two directions. It takes 4 protons to cap one side or 8 protons to cap both sides of a nuclet. When both sides are capped it is called a neutral ending and that part of the atom is no longer chemically reactive. The capping protons are always colored green.
Atoms are built by putting together nuclets in a tree like fashion. For small elements there are few options and they can only grow one way. As elements get bigger they have multiple branches that can grow simultaneously and so the options for how it will grow become more complex. However nature likes to keep things in equilibrium and it works to complete the nuclets on all branches before starting a new growth cycle.
Magically, or is it coincidentally, or maybe mysteriously, or how about convincingly - it just so happens when all branches have been capped, the number of protons equals that of one of the noble gases. It seems highly likely this is coincidental. It is one of the easier to understand correlations between SAM and the elements.
The isotopes are also explained by SAM but that will be coming later. Right now we are focusing on the most abundant isotopes of each element.
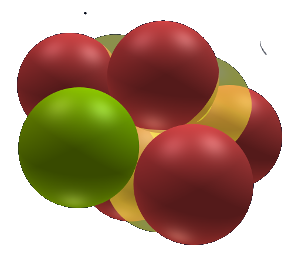
Helium 4
Atomic structure
N0:
state: helium
protons:
P0:
P1:
P2:
P3:
electrons:
E01: {protons: [P0, P3]}
E02: {protons: [P1, P2]}
This is helium a tetrahedron
Neon 20
Atomic structure
N0:
state: initial
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P10:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
electrons:
E01: {protons: [P16, P17]}
E02: {protons: [P18, P19]}
E03: {protons: [P12, P13]}
E04: {protons: [P14, P15]}
E05: {protons: [P0, P1]}
E06: {protons: [P9, P0]}
E07: {protons: [P8, P5]}
E08: {protons: [P4, P1]}
E09: {protons: [P10, P3]}
E10: {protons: [P11, P7]}
E11: {protons: [P6, P2]}
This is a depiction of Neon
Argon 38
Atomic structure
N0:
state: final
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P10:
P11:
P12:
P13:
P14:
P15:
neutrons: [U00]
outerIcosaFaces: []
nuclets:
N01:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
neutrons: [U00, U10]
outerIcosaFaces: []
This is Argon
Krypton 80

Atomic structure
N0:
state: final
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P10:
P11:
outerIcosaFaces: []
nuclets:
N00:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P16:
P17:
P18:
P19:
outerIcosaFaces: []
nuclets:
N000:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
outerIcosaFaces: []
N01:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P16:
P17:
P18:
P19:
outerIcosaFaces: []
nuclets:
N010:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
outerIcosaFaces: []
This is Krypton
Xenon 126

This is Xenon
Radon 222
Atomic structure
N0:
state: final
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P10:
P11:
nuclets:
N00:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
nuclets:
N000:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
nuclets:
N0000:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
neutrons: [U00]
N0001:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
neutrons: [U00, U10]
N001:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
nuclets:
N0010:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
neutrons: [U00, U10]
N0011:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
N01:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
nuclets:
N010:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
nuclets:
N0100:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
neutrons: [U00]
N0101:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
N011:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P16:
P17:
P18:
P19:
neutrons: [U10]
nuclets:
N0110:
state: final
attachAngle: 3
protons:
P0:
P1:
P2:
P3:
P4:
P5:
P6:
P7:
P8:
P9:
P11:
P12:
P13:
P14:
P15:
P16:
P17:
P18:
P19:
This is radon